Telomeres: A Comprehensive Guide to Cellular Longevity, Protection, and Health Optimization
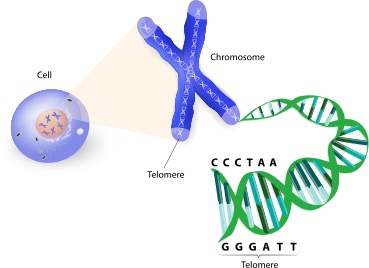
Telomeres are specialized nucleoprotein structures located at the terminal ends of chromosomes. Their primary function is to safeguard genetic information, preventing genomic degradation during cell division. The existence of these protective caps was first identified by Hermann Muller and Barbara McClintock through their respective studies on Drosophila and maize. They observed a unique terminal structure that prevents cells from identifying chromosome ends as damaged DNA, thereby inhibiting abnormal end-to-end fusion or enzymatic degradation.
In 2009, the scientific significance of telomeres was solidified when Elizabeth Blackburn, Carol Greider, and Jack Szostak were awarded the Nobel Prize in Physiology or Medicine for discovering how chromosomes are protected by telomeres and the enzyme telomerase. This breakthrough catalyzed extensive global research into cellular aging and longevity.
Molecular Architecture: The T-Loop and Repetitive Sequences
In vertebrates, including humans, telomeres consist of non-coding, highly repetitive DNA sequences—specifically “TTAGGG.” These sequences can span hundreds to thousands of repeats. The terminal end of the telomere features a single-stranded G-rich 3′ overhang that folds back upon itself to form a large circular structure known as the Telomere loop (T-loop). Acting much like a molecular “paperclip,” this structure is stabilized by a specialized protein complex that binds specifically to the TTAGGG sequence, ensuring the stability of the chromosomal terminus.
The “End Replication Problem” and the Hayflick Limit
DNA replication is subject to a fundamental mechanical constraint known as the “End Replication Problem.” As described by Olovnikov, the action of DNA polymerase (the enzyme responsible for synthesizing new DNA strands) can be likened to a train moving along a track. Just as a train cannot lay track directly beneath its own wheels, DNA polymerase cannot replicate the very beginning of a DNA strand.
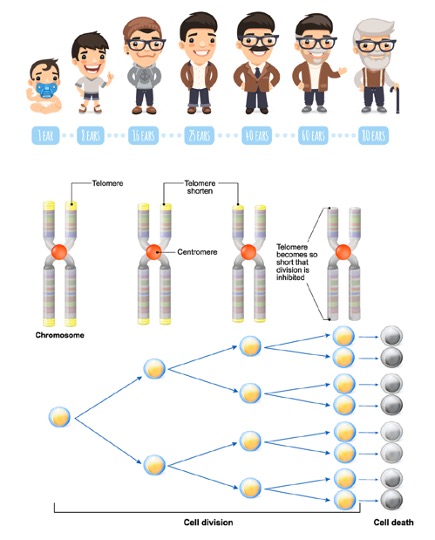
Consequently, vital genetic information would be lost with every cell division if not for telomeres. These non-coding buffers absorb the attrition, shortening by approximately 30–200 base pairs per replication cycle. When telomeres reach a critically short threshold, the cell reaches the Hayflick Limit, triggering either cellular senescence (permanent growth arrest) or apoptosis (programmed cell death).
Telomerase: The Enzyme of Cellular Immortality
Telomerase is a ribonucleoprotein enzyme that addresses the end replication problem by systematically extending telomere length. Under physiological conditions, telomerase is highly active during early embryonic development and within adult stem cells. However, it can also become aberrantly activated in cancer cells, granting them the capacity for indefinite proliferation. In contrast, most healthy somatic cells produce negligible or no telomerase, making them subject to the natural aging process.
Oxidative Stress: A Catalyst for Telomeric Attrition
Oxygen Free Radicals (such as $O_2^-$, $H_2O_2$, and Nitric Oxide) are metabolic byproducts generated primarily within the mitochondria during energy production. While some radicals assist in pathogen defense, an excess can lead to Oxidative Stress. These unstable molecules “steal” electrons from DNA, proteins, and lipids to achieve stability, triggering a destructive chain reaction.
Antioxidants counteract this by donating electrons to free radicals without becoming unstable themselves. An imbalance between free radicals and antioxidant defenses is a primary driver of accelerated telomere shortening. Research indicates that high oxidative stress levels cause telomeres to degrade significantly faster than the rate predicted by the end replication problem alone. The GGG triplet in telomeric DNA is particularly vulnerable to oxidative damage, alkylation, and UV radiation. Chronic oxidative stress is also a hallmark of inflammatory diseases and is prevalent in patients with coronary artery disease, Type 2 diabetes, and Chronic Obstructive Pulmonary Disease (COPD).
Micronutrients and Protective Interventions
Evidence suggests that Micronutrients—specifically antioxidant vitamins and minerals—can mitigate oxidative stress and chronic inflammation, thereby slowing telomere attrition. Clinical studies have shown that women who regularly consume multivitamins tend to possess longer telomeres compared to control groups. Key nutrients associated with telomere maintenance include:
Folate and Vitamin B12
Vitamins A, D, C, and E
Nicotinamide
Omega-3 Fatty Acids
Telomere Length as a Clinical Biomarker
Telomere shortening is intrinsically linked to cellular aging. At birth, telomeres typically measure approximately 10,000 base pairs (bp), progressively decreasing with age. Leukocyte telomere length (LTL) has emerged as a significant predictor for age-related morbidities, including atherosclerosis, myocardial infarction, Alzheimer’s disease, hypertension, and diabetes.
Notably, centenarians (those living over 100 years) often exhibit longer-than-expected telomeres. Conversely, individuals with shorter LTL face an increased risk of cancer and higher cancer-related mortality rates. Because telomere length reflects both genetic predisposition and lifestyle impacts, it serves as a powerful biological age biomarker, offering a more accurate assessment of health risks than chronological age alone.
Conclusion and Recommendations
To effectively manage the rate of biological aging, annual telomere length screening is recommended. This allows for the monitoring of attrition rates and the implementation of personalized therapeutic strategies, including:
Optimizing sleep hygiene
Regular physical exercise
Effective stress management
Targeted nutritional supplementation
By proactively protecting telomere integrity, individuals can significantly enhance their long-term health trajectory and reduce the risk of age-related diseases.
References
Blackburn, E. H. “Telomeres and Telomerase: The Means to the End (Nobel Lecture).” Angew Chem Int Ed Engl 49, no. 41 (Oct 04 2010): 7405-21.
de Vos-Houben, J. M., N. R. Ottenheim, A. Kafatos, B. Buijsse, G. J. Hageman, D. Kromhout, and E. J. Giltay. “Telomere Length, Oxidative Stress, and Antioxidant Status in Elderly Men in Zutphen and Crete.” Mech Ageing Dev 133, no. 6 (Jun 2012): 373-7.
Lapham, K., M. N. Kvale, J. Lin, S. Connell, L. A. Croen, B. P. Dispensa, L. Fang, et al. “Automated Assay of Telomere Length Measurement and Informatics for 100,000 Subjects in the Genetic Epidemiology Research on Adult Health and Aging (Gera) Cohort.” Genetics 200, no. 4 (Aug 2015): 1061-72.
Lin, J., E. Epel, and E. Blackburn. “Telomeres and Lifestyle Factors: Roles in Cellular Aging.” Mutat Res 730, no. 1-2 (Feb 01 2012): 85-9.